Introduction: Human leukocyte antigen (HLA) plays a major role in the interaction between the immune system and oncogenic process in various types of tumors. Emerging data suggests that the HLA system may have profound effects on response to immunotherapy and other types of therapy. Structural defects in HLA genes and variation in methylation have been reported to play a role in the expression of various HLA-I and HLA-II genes. Furthermore, an association between the expression of specific HLA-I or HLA-II genes and prognosis and outcome has been reported in some types of leukemia. Since HLA-I is expressed on all cells while HLA-II is expressed mainly in antigen presenting cells, we hypothesized that profiling the expression levels of both HLA-I and HLA-II in the bone marrow of patients with various myeloid neoplasms may provide information on the interaction between the microenvironment and the leukemic cell in these diseases. We used next generation sequencing to evaluate the expression level of HLA-I and HLA-II genes in myeloid neoplasms and compared these levels to normal bone marrow control. We used targeted next generation sequencing (NGS) for quantification of the RNA expression of 15 HLA-I and HLA-II genes.
Methods: RNA was extracted from fresh bone marrow aspiration samples from 115 acute myeloid leukemia (AML), 196 clonal hematopoiesis of indeterminate potential (CHIP), 84 myeloproliferative neoplasm (MPN), 177 myelodysplastic syndrome (MDS), 40 acute lymphoblastic leukemia (ALL), and 279 normal bone marrow cases. RNA sequencing was performed using a targeted hybrid capture panel. The sequenced HLA-I genes were HLA-A, HLA-B, HLA-C, HLA-E, HLA-F, and HLA-H. The sequenced HLA-II genes were: HLA-DPA1, HLA-DPB1, HLA-DQA1, HLA-DQB1, HLA-DQB2, HLA-DRA, HLA-DRB1, HLA-DRB5, and HLA-DRB6. Salmon v1.4.0 software was used for expression quantification (TPM). To evaluate the depth of the significance in variation of the expression profiles between diseases we tested machine learning algorithm (random forest) incorporating the expression of all 15 genes in blindly classifying diseases. Two thirds of samples were used for training the random forest algorithm and one third was used for testing.
Results:
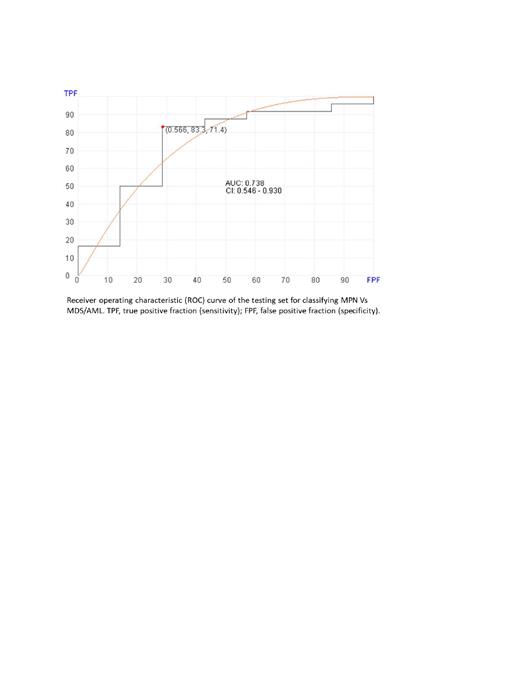
Bone marrow samples with CHIP showed no significant difference from normal bone marrow in level of expression of HLA-I genes but showed significantly (P <0.001) lower level of most HLA-II markers (DMB, DQA1, DQB2, DRB1, and DRB5). The same pattern was observed in MDS samples. In contrast, AML showed significant (P < 0.001) increase in the expression of both HLA-1 genes (A, C, and F) and HLA-II genes (DQA1, DPA1, DQB1, DRB6, and DPB1). Bone marrow samples from ALL patients showed significantly (P< 0.001) higher expression levels in DRB1 and DQA1 as compared with normal bone marrow. MPN samples showed no statistically significant difference from normal BM in any of the HLA-I or HLA-II genes, after adjusting for multiple testing. Machine leaning algorithm poorly classified MDS and AML and suggests complex overlap between the two diseases in HLA response. In contrast, the machine learning algorithm showed significant difference between MPN, MDS, and AML, and ability to distinguish all MPN cases with AUC of 0.738 (95% CI : 0.546-0.930) in the testing set (see figure).
Conclusions:
These findings show that the bone marrow microenvironment immune profile in MDS and CHIP likely plays a major role in the disease. Furthermore, this data suggests that this immune microenvironment is critical in CHIP and perhaps that therapeutic approaches supporting this immune microenvironment may help in preventing CHIP from progressing to MDS. The data also supports that AML is a disease that completely overwhelms the HLA immune system. In contrast, MPN appears to evade the HLA system and perhaps activating this system may help in the therapy of MPN.
Disclosures
Ip:Genomic Testing Cooperative: Current equity holder in private company; Seattle Genetics: Speakers Bureau; AstraZeneca: Speakers Bureau; Secura Bio: Consultancy, Membership on an entity's Board of Directors or advisory committees; TG Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; MJH Life Sciences: Honoraria; COTA: Current holder of stock options in a privately-held company; Merck: Current holder of stock options in a privately-held company. McCloskey:Takeda: Speakers Bureau; Stemline Therapeutics: Speakers Bureau; Jazz Pharmaceuticals: Speakers Bureau; Incyte: Speakers Bureau; Amgen: Speakers Bureau; BluePrint Health: Speakers Bureau; Novartis: Consultancy; Blueprint Medicines: Consultancy; Bristol-Myers Squibb/Pfizer: Consultancy, Honoraria, Speakers Bureau; BluPrint Oncology: Honoraria. Koprivnikar:Apellis: Consultancy; Novatis: Consultancy; GSK: Consultancy; Alexion: Consultancy. Goy:Bristol Myers Squibb/Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; MorphoSys: Research Funding; Hoffman la Roche: Consultancy, Honoraria, Research Funding; Genentech: Research Funding; Seagen: Research Funding; Pharmacyclics LLC, an AbbVie Company: Other: Steering Committee, Research Funding; Infinity: Research Funding; OMI: Current Employment; Karyopharm: Research Funding; Practice Update Oncology: Consultancy, Honoraria; Resilience: Current holder of stock options in a privately-held company; Genomics Testing Cooperative LLC: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees, Other: Leadership Role; Novartis: Consultancy, Honoraria; Acerta: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Other: Steering Committee, Research Funding; Medscape: Consultancy; Kite, a Gilead Company: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Steering Committee, Research Funding; Clinical Advances in Hematology & Oncology: Consultancy; AbbVie/ Pharmacyclics LLC: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Steering Committee, Research Funding; Regional Cancer Care Associates, OMI: Current Employment, Research Funding; Alloplex: Current holder of stock options in a privately-held company, Honoraria, Membership on an entity's Board of Directors or advisory committees; Michael J. Hennessey: Consultancy, Honoraria; COTA Healthcare: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees, Other: Leadership Role; Xcenda: Consultancy, Honoraria; OncLive Peer Exchange: Honoraria; Constellation: Research Funding; Vincerx: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other; Physicians Education Resource, LLC: Consultancy, Honoraria, Other: travel, accommodations, and expenses; Verastem: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal